The ANSM affirms its desire to see a significant reduction in the use of mercury-based amalgams to treat cavities. It issues new recommendations to dentists.

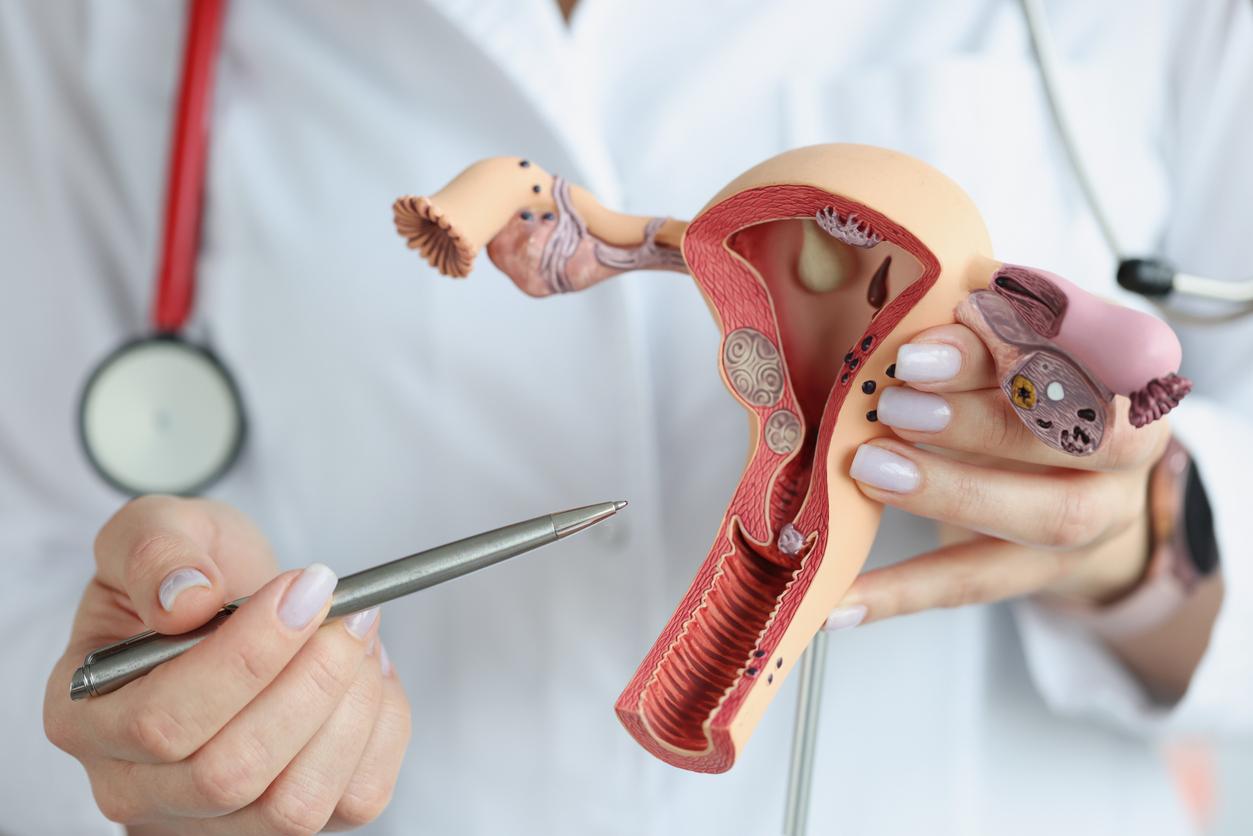
This is a warning to dentists that the Drug Safety Agency formulated yesterday. In a press release published on Thursday, the ANSM affirms its desire to see a significant reduction in the use of mercury-based amalgams in the treatment of dental caries. These devices are in fact suspected of being toxic for the nervous, immune, hormonal and reproductive systems.
Very limited clinical uses
As such, it issues recommendations for dentists in order to specify the limited clinical situations in which amalgam can still be used. This is the case, for example, with restoration of permanent posterior teeth (molars and premolars) in the event of high caries prevalence and multiple and extensive lesions.
Conversely, dental amalgams should not be used in patients with a proven history of allergy to mercury and identified by patch tests.
Dental amalgams are, as a precaution, also contraindicated in patients whose kidneys are weakened by a history of impaired renal function, adds the ANSM.
Limit the concentration of mercury in the air of the practices
At the same time, dentists are also reminded of the importance of respecting the rules of oral hygiene when handling these devices. Dental amalgams should therefore be used in pre-dosed capsule packaging. Hygiene rules which must be respected “in order to limit as much as possible the concentration of mercury in the atmosphere of dental offices”, specifies the Agency.
Report any adverse reactions
Finally, the ANSM wishes to make both healthcare professionals and patients aware of the importance of reporting any undesirable effects in relation to the use of a filling material. The Agency also recalled, during the risk prevention committee of October 2014, the need to promote research in the field of biomaterials in order to broaden the offer of alternative solutions to dental amalgams. available.
.