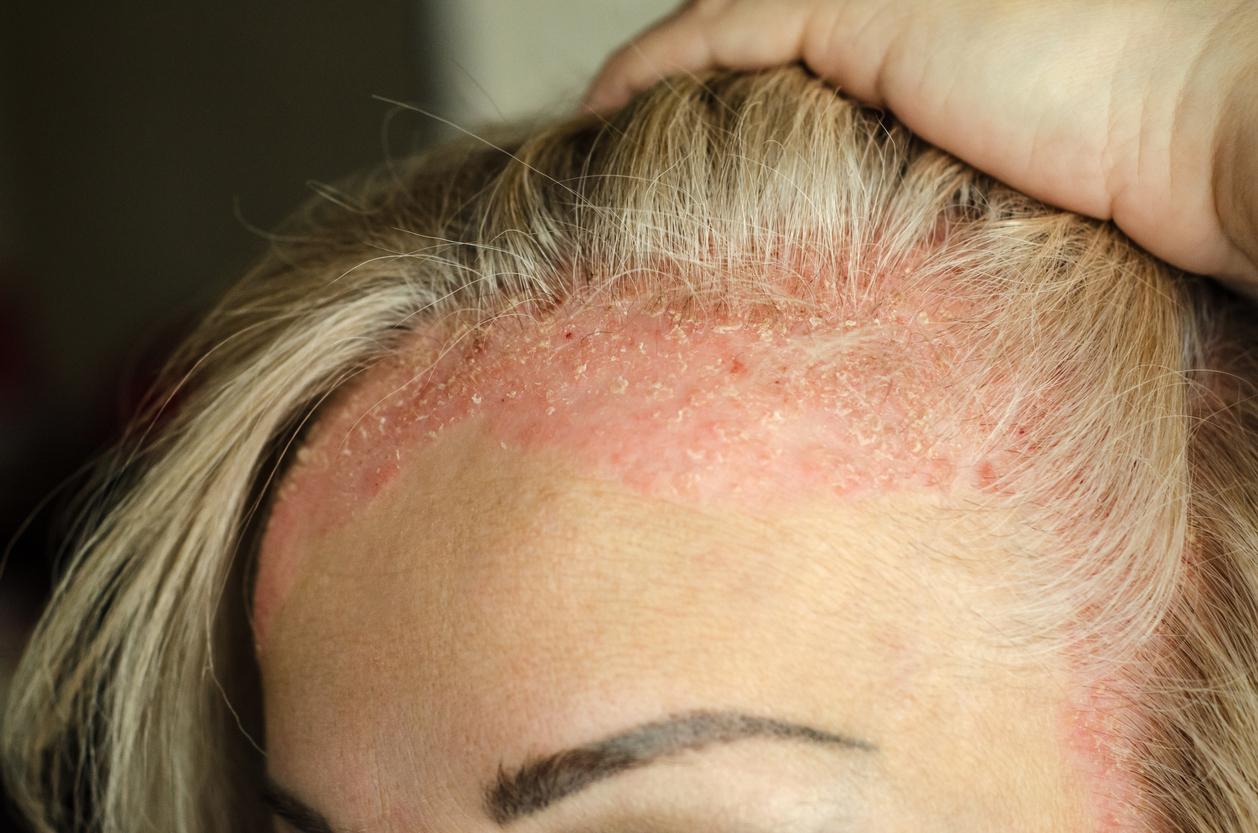
Acitretin is, marketed in France under the name Soriatane, is a retinoid (derivative of vitamin A) since 1988. It is indicated in severe forms of psoriasis as a single treatment or in combination with puvatherapy. Its prescription is very framed to protect women from malformations during pregnancy.
A heavy treatment
Like all systemic retinoids, acitretin is a powerful teratogen (which can create malformations in children) justifying specific preventive measures.
The ANSM reminds that health professionals must imperatively inform themselves and ensure that their patients are on effective contraception for the entire duration of treatment and the 2 years following its discontinuation.
They must also prescribe a pregnancy test within the 3 days preceding each monthly prescription, then 2 months after stopping treatment and during the 2 years following this stop on the dates they have agreed.
The ANSM also confirms that pharmacists cannot deliver this treatment without mentioning the negative pregnancy test recorded on the health record, which is compulsory for each consultation and for each purchase.
Patients should not consume alcohol during treatment and for 2 months after its discontinuation.
An alarming record
However, a study carried out between 2007 and 2012 among 7,663 women aged 15 to 49 initiating treatment with acitretin reveals an alarming assessment of compliance with safety measures.
Indeed, the realization of the pregnancy test was observed in only 11% of cases. During treatment, nearly two-thirds of women (65%) did not undergo any pregnancy test; after stopping treatment, 4 out of 5 women (77%) did not take any pregnancy tests within two years of stopping treatment.
In addition, 357 pregnancies were observed during the treatment or in the two years following its discontinuation, of which nearly 40% were terminated (medical termination of pregnancy, voluntary termination of pregnancy or spontaneous miscarriage). For 53 of these pregnancies, the delivery of acitretin took place during the pregnancy.
In this context, the ANSM recalls “the importance of properly informing all women of childbearing age that a treatment by Soriatane is incompatible with pregnancy, throughout the duration of treatment as well as for the 2 years following this treatment, due to the risk of storage in fat of a teratogenic metabolite, the formation of which is favored by the consumption of alcohol” and put in place new measures. The initial prescription will be reserved for dermatologists and can only be renewed for one year after which only this specialist can again issue a prescription.