The lawyer for patients engaged against the Merck laboratory, producer of Levothyrox, reveals that the old formula of the drug would still be produced in France.
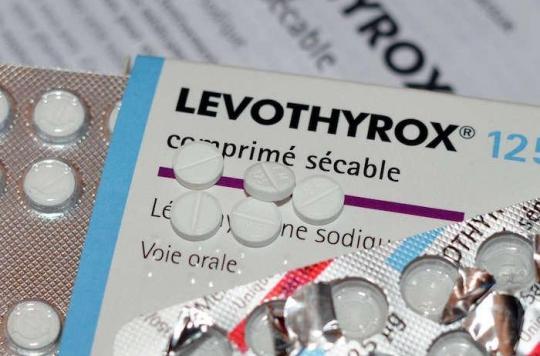
New twist in the Lévothyrox affair. The lawyer for the patients who have taken legal action against the laboratory affirms that the old formula of the drug would still be produced in France, in Isère. But the drugs would be intended exclusively for the Italian market.
Italian preference
It is the lawyer Maître Christophe Lèguevaques who is responsible for defending the patients. He explains in a press release: “We let patients suffer while a solution exists in Ise? Re!” We want to know why the French health authorities did not require? of Merck and its subcontractor that the production of the old formula be sold on the market? French. […] We want to know the production capacities of the Bourgoin-Jallieu site, to know if they can be increased and in what proportions. We want to know why, faced with the multiplication of cases of side effects caused by the new formula, Merck did not order? at? its subcontractor to increase its production of the original Le? vothyrox. “
“A different version of the drug”, justifies Merck
Maître Christophe Lèguevaques sent by bailiff an “interpellative summons” to the Minister of Health Agnès Buzyn, to the Merck laboratory, to the company Panthéon (the laboratory which manufactures the drug in Isère) and to the National Security Agency of the Drug. A provision which makes it possible to directly question a person or an entity in order to obtain answers.
For the laboratory and the Ministry of Health, the formula intended for the Italian market is different from the old French version of the drug. Also, it does not meet the same security requirements and the same procedures.
The Levothyrox affair began last October with complaints from patients en masse. The new formulation of Levothyrox, a drug that treats thyroid disorders, was released last March. Very quickly, some patients felt strong side effects.
.















-1597155771.jpg)