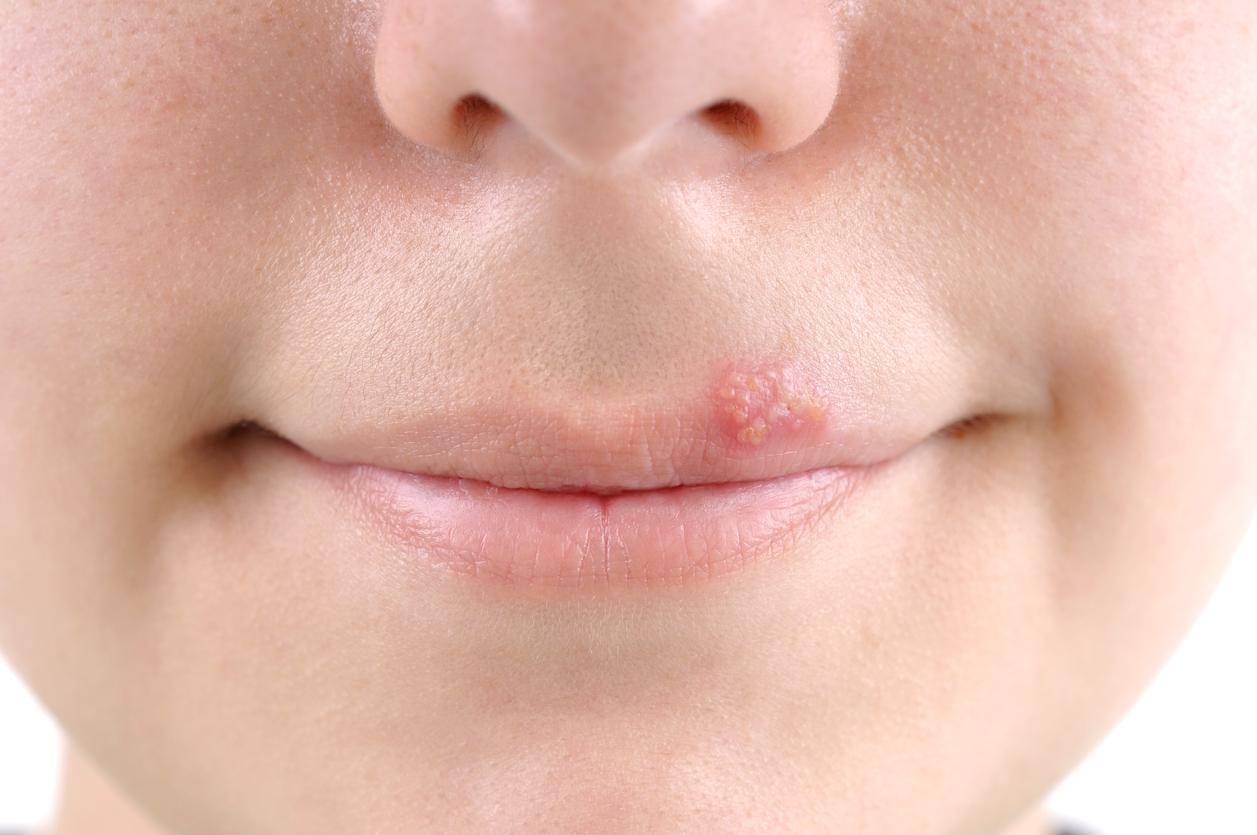
The American Medicines Agency has authorized the marketing of a test capable of detecting the herpes virus in a simple blood sample taken from the fingertip.
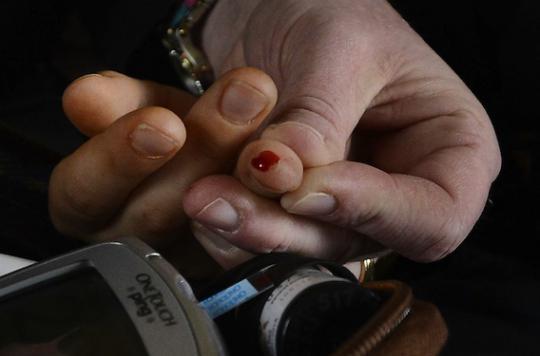
A start-up in Silicon Valley received this Thursday, the approval of the United States Medicines Agency (FDA) for its test detecting the herpes virus from a simple prick of the fingertip.
The company Theranos, based in Palo Alto in California (United States), has directed all its research towards the development of rapid and inexpensive tests. The FDA-cleared test, named HSV-1, would cost less than $ 10 (about $ 9), according to the company.
This works on the same principle as the current blood test. In a blood sample, the test looks for the presence of antibodies directed against the virus Herpes simplex type 1 (HSV-1), responsible for the famous “cold sore”, but also for genital herpes.
To obtain this clearance, Theranos provided the FDA with test results from 818 men and women.
A first step towards transparency
“In order to implement our vision of early detection, laboratory tests must meet the highest quality criteria – standards set by the FDA,” explained in a communicated Elizabeth Holmes, Founder and CEO of Theranos.
The Agency review is the only rigorous process we wanted to go through because we are committed to ensuring that our systems and tests are of high quality, and that patients and physicians have access to the most accurate information. concerning their health. We look forward to continuing our strong relationship with the Agency. “
An announcement that sounds like a settling of scores. According to Washington post, the company is regularly accused of lacking transparency. They mention in particular an editorial signed by John Ioannidis, president of the prevention department at Stanford University, in the journal JAMA in February. FDA clearance should lift the veil on some of the company’s practices, he said.
.