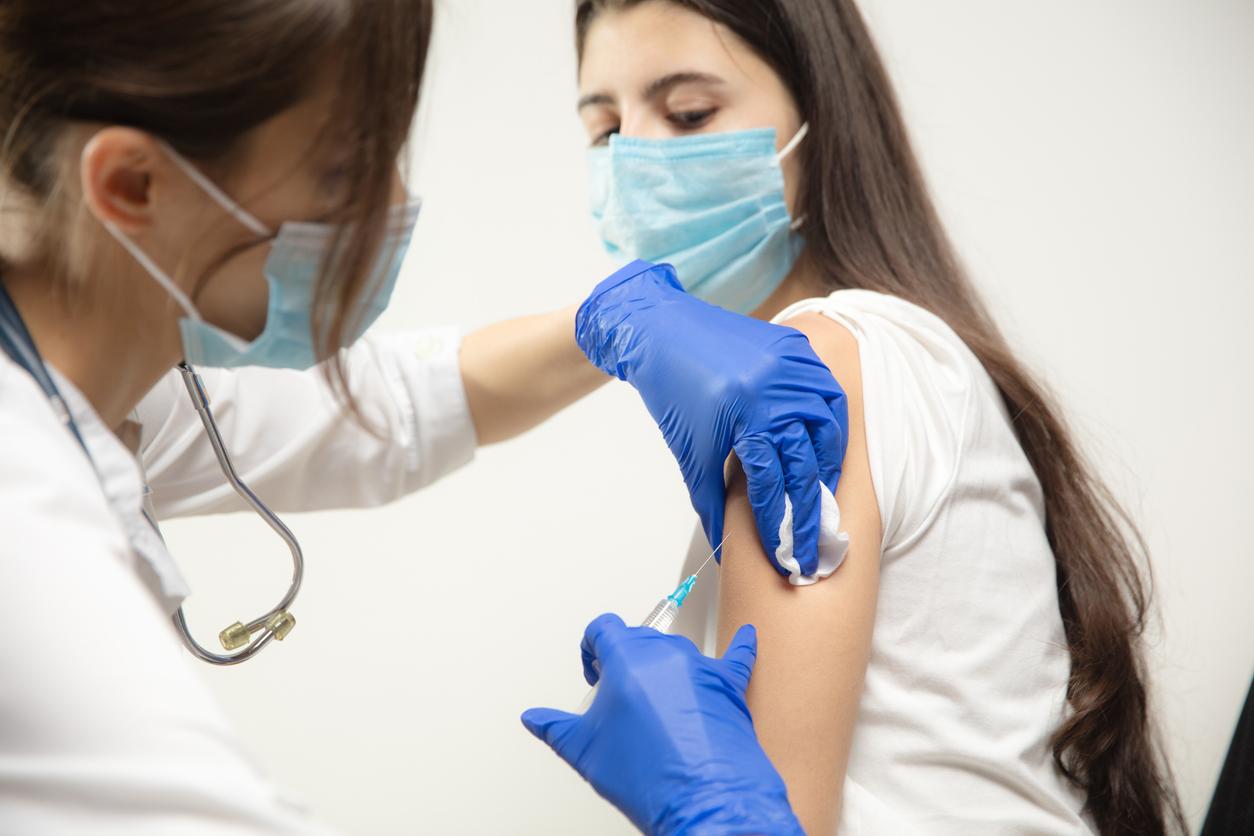
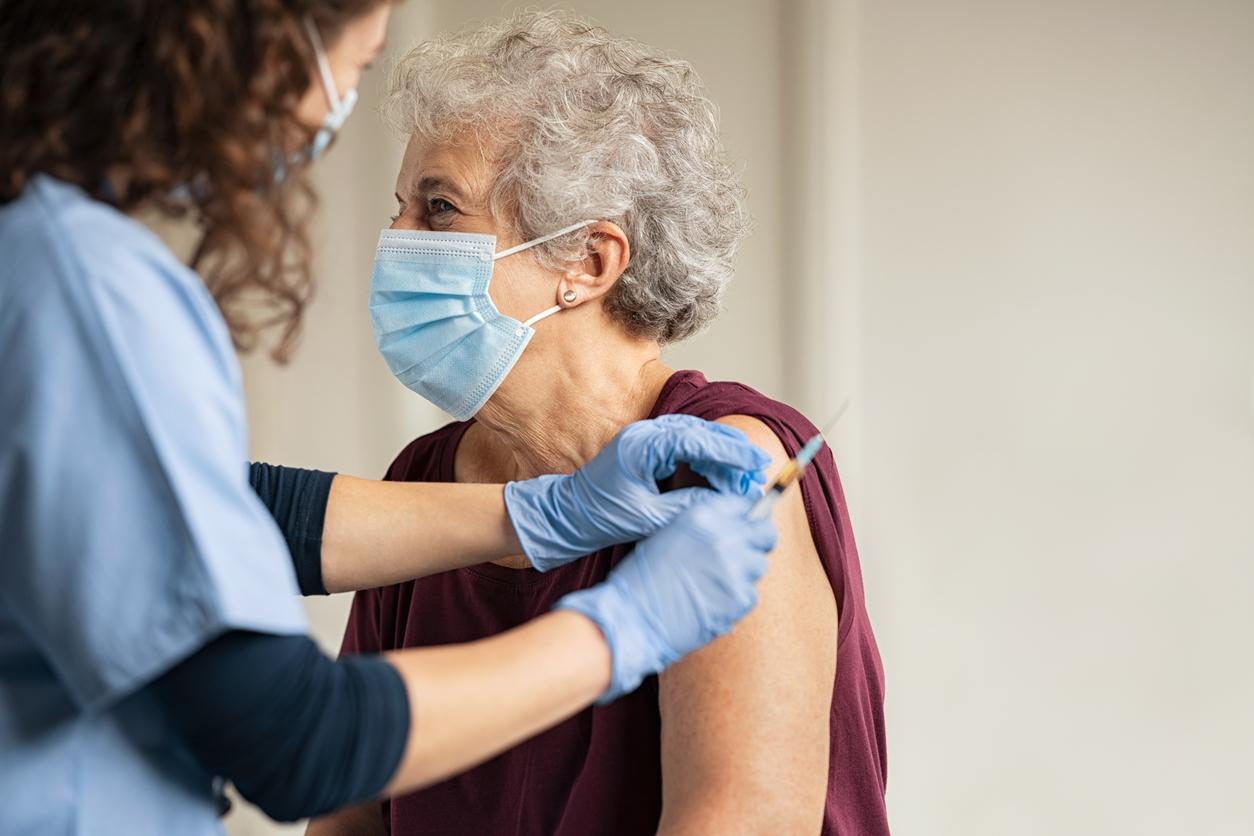
Pharmacists from Auvergne-Rhône-Alpes and Nouvelle-Aquitaine are now authorized to vaccinate against influenza. An experimental measure which should last three years.

The 2017-2018 vaccination campaign against seasonal influenza is open. For the winter of 2016-2017, less than one in two French people (46%) had been vaccinated. Insufficient coverage to protect the most vulnerable people. The French Ministry of Health and Public Health are trying to increase it, with different strategies.
This year, the regional health agencies (ARS) of the Auvergne-Rhône-Alpes and Nouvelle-Aquitaine regions are launching an experiment, for three years. Pharmacists will be able to vaccinate people who present with a vaccination voucher or a prescription from their doctor. Until now, the act of vaccination was not allowed to them.
A third of the pharmacists involved
“In Auvergne-Rhône-Alpes, around 2,000 pharmacists have registered with the ARS, including 500 in the Rhône department”, reports Véronique Nouri, president of the Federation of Pharmaceutical Unions of the Rhône (FSPF69), a department which counts 585 pharmacies. This represents about a third of the professionals in the department.
The pharmacists interested in the experiment had to follow a one-day training course, in order to be informed on the regulatory details, in particular on the material conditions of reception of the patients, who will be received in a closed room, isolated from the pharmacy.

Doctors also reminded them of the vaccination gesture and the recommendations for questioning. Pharmacists will have to question patients about their allergies (to eggs and antibiotics in particular). They should also ask them if they have ever had a flu shot.
Because it is one of the conditions of the authorization to vaccinate. Pharmacists can only give the injection to people who have already been immunized in the past, during a previous campaign.
A first step
For now, interest is meager for pharmacists, and even financially counterproductive. The ARS will pay compensation of 150 euros to those who have made at least five injections during the season. “The procedure is not done in 2 minutes,” says Véronique Nouri. Experimentation isn’t profitable for us, but that’s not why we wanted to get involved. This is not a first step ”.
For pharmacists, this empowerment in the vaccine act was indeed a long-standing demand. While the experimentation carried out by the two regions serves immunization coverage, it also serves to test the effectiveness of the involvement of pharmacy professionals. “In Canada or in the United States, they are authorized to carry out all the vaccines”, continues Mrs. Nouri.
A little more waiting
The president of the FSPF69 is therefore delighted with this new perspective. However, she regrets the target designated by Health Insurance. Vaccination vouchers are sent to people considered at risk, and prescriptions are only given to people who go for a consultation. They therefore exclude healthy young people and workers from the promotion of vaccination. “To protect the most vulnerable people, vaccination coverage should exceed 75%,” she explains. For that, it would be necessary to include this population ”.
For others, with a pass, it is now possible to go to the pharmacy. But Véronique Nouri recommends to wait a little longer. “People vaccinated in early October will no longer be immunized in March, and the flu season has not yet started,” she says.
During the winter of 2016-2017, the flu would have affected around 2.5 million people in France, according to figures from Public Health France. The health agency estimates that 14,400 deaths could be attributed to him.
The influenza virus, champion of mutation
There are three different strains of influenza: A, B and C. Strains A and B are the most common, and these are the ones the vaccine is produced against. It must be renewed every year. “The influenza virus is the champion of the mutation, explains for Supplement Prof. Bruno Lina, virologist and director of the national influenza reference center. It is its nature which means that, each year, a new strain is responsible for the epidemic which affects several million people in France. “
The vaccine is made from inactivated viruses from previous years, and in particular the latest one. The World Health Organization is following the evolution of mutations and providing its recommendations for the vaccine for the coming winter.
This year it contains three strains. Two type A strains, A / Michigan / 45/2015 (H1N1) pdm09, and A / Hong Kong / 4801/2014 (H3N2), as well as one type B strain, B / Brisbane / 60/2008.
However, the efficacy of the vaccine is not guaranteed. Production takes time, about six months. “If the virus changes in the meantime, it is too late to adapt the vaccine,” adds Professor Lina. This is what happened in 2014: a late epidemic occurred in April-May, when manufacturing had already started. The vaccine for the following season was then lacking in effectiveness. On average, and depending on the year, it is between 50 and 70%.
Find the Supplement
Vaccination: put an end to doubts
.