According to US health authorities, out of nearly 12.5 million injected doses of this vaccine, 100 people have developed Guillain-Barré syndrome. Of these 100 identified cases, 95 were in serious condition and were hospitalized. One person died. “The chances of this happening are very low and the rate of reported cases only exceeds the baseline rate in the general population by a small margin.”, nevertheless indicated Monday evening the pharmaceutical company Johnson & Johnson in a press release.
Urgently approved in the United States last February in the United States and available in France since April, the single-dose Jensen vaccine had already been suspended for 10 days following cases of thrombosis.
What is this syndrome?
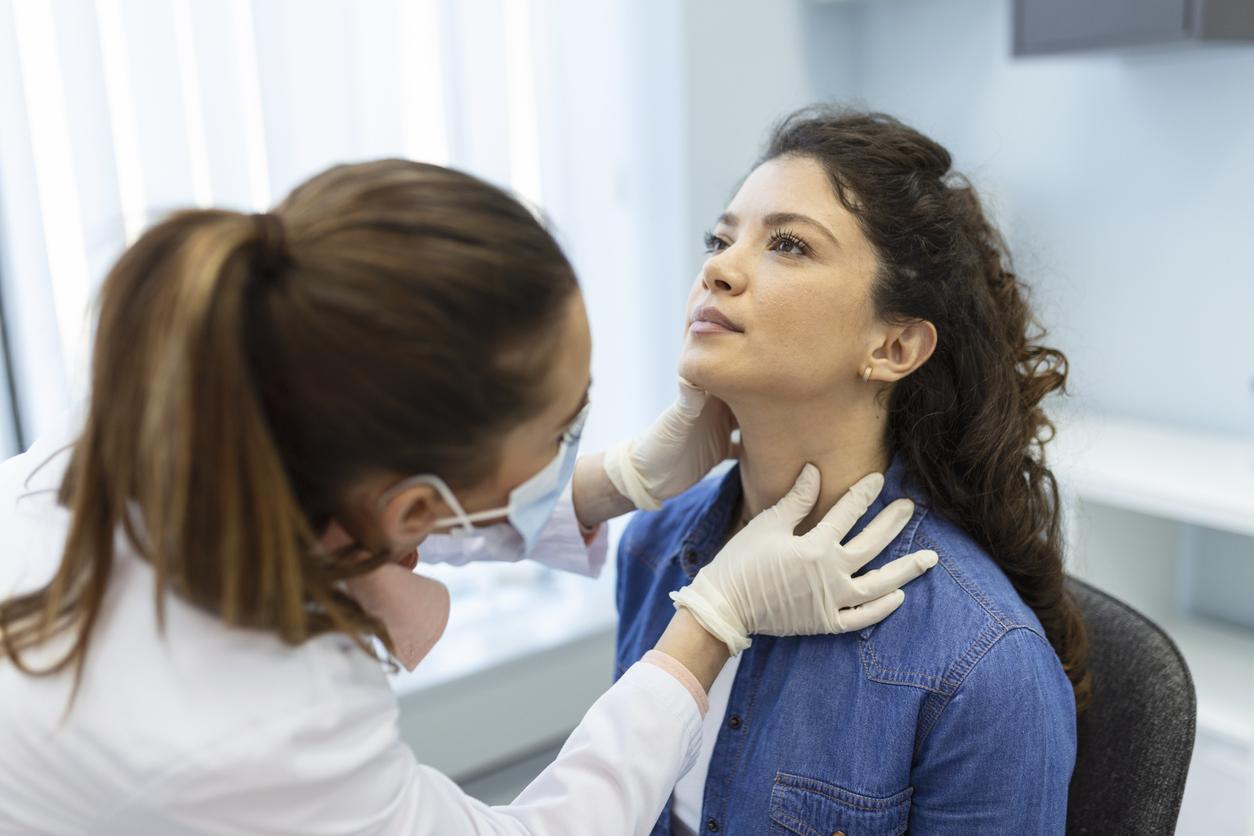
Guillain-Barré syndrome (GBS), or Acute Inflammatory Polyradiculoneuritis, is an autoimmune disease that causes peripheral nerve damage and paralysis. This paralysis is said to be extensive because it generally begins with the legs and arms and then spreads to the rest of the body. This syndrome would affect between 3,000 and 6,000 people each year in the United States.
In its statement released on July 12, the FDA specifies that in the majority of individuals, symptoms can appear within 42 days of receiving the viral vector vaccine. People who have received the American serum should see a doctor or go to the emergency room immediately if: tingling and strange sensations in the feet and hands, difficulty swallowing or chewing, or if your vision becomes weak or if you are unable to move your eyes.