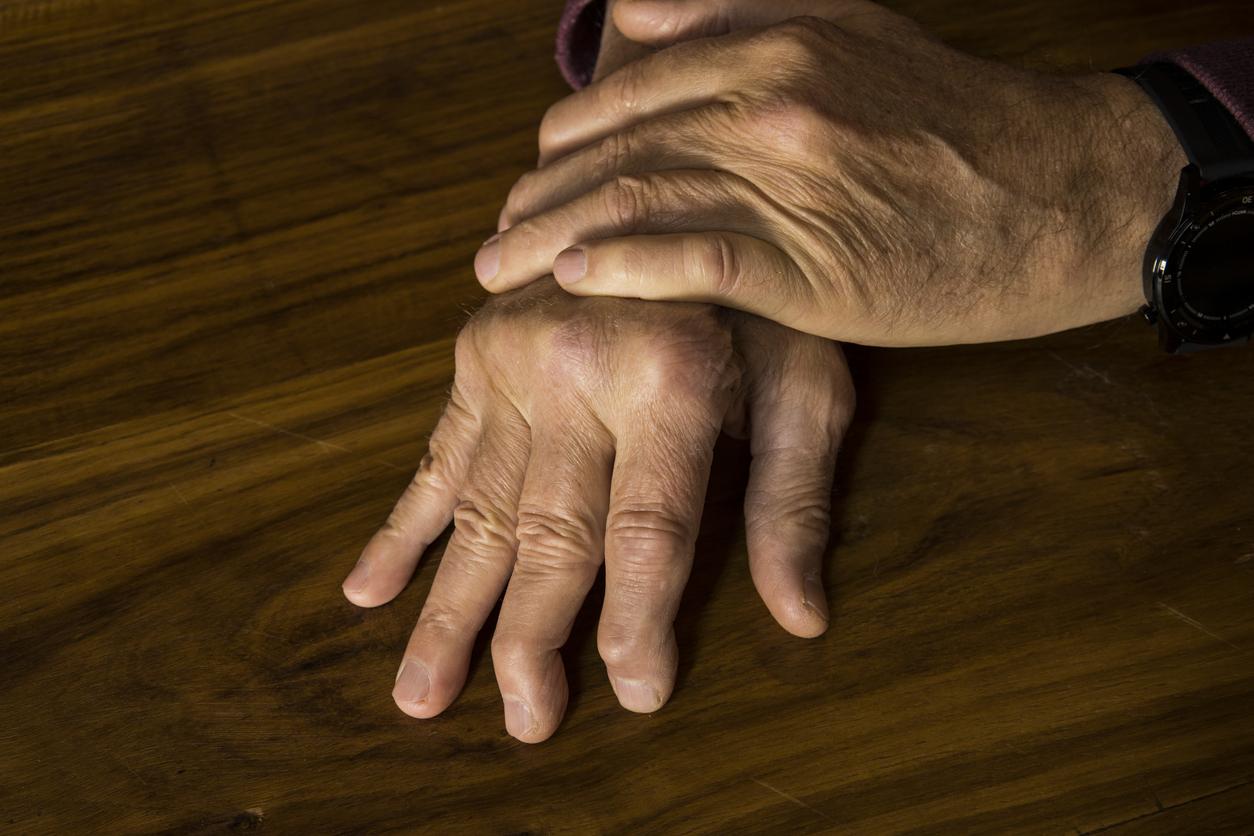
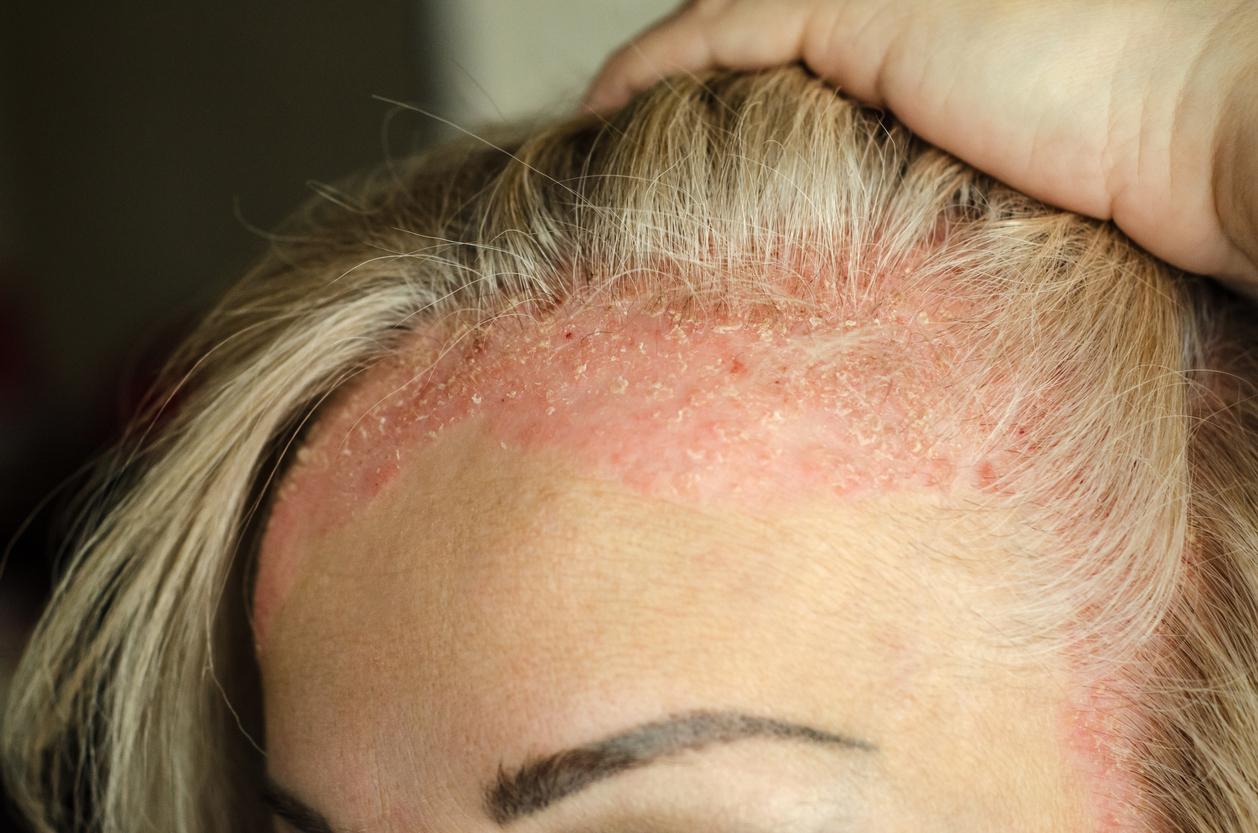
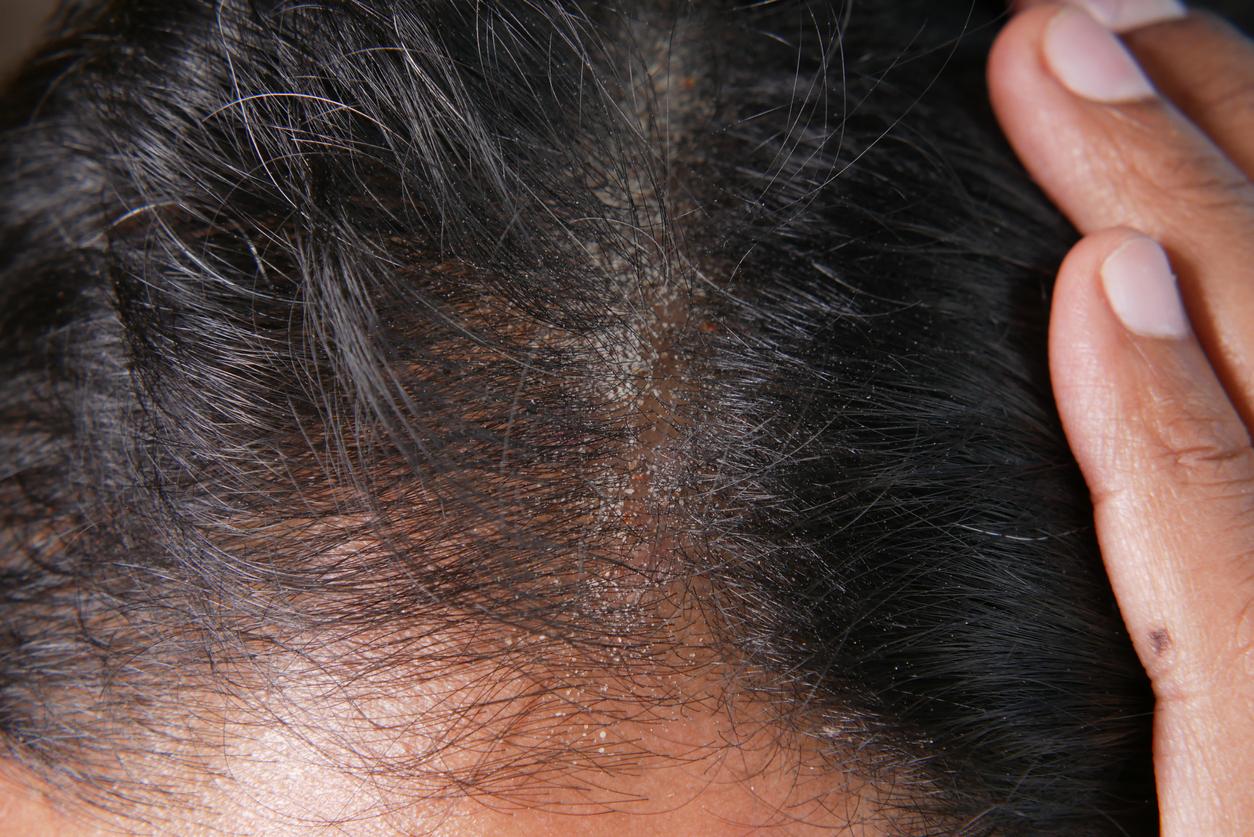
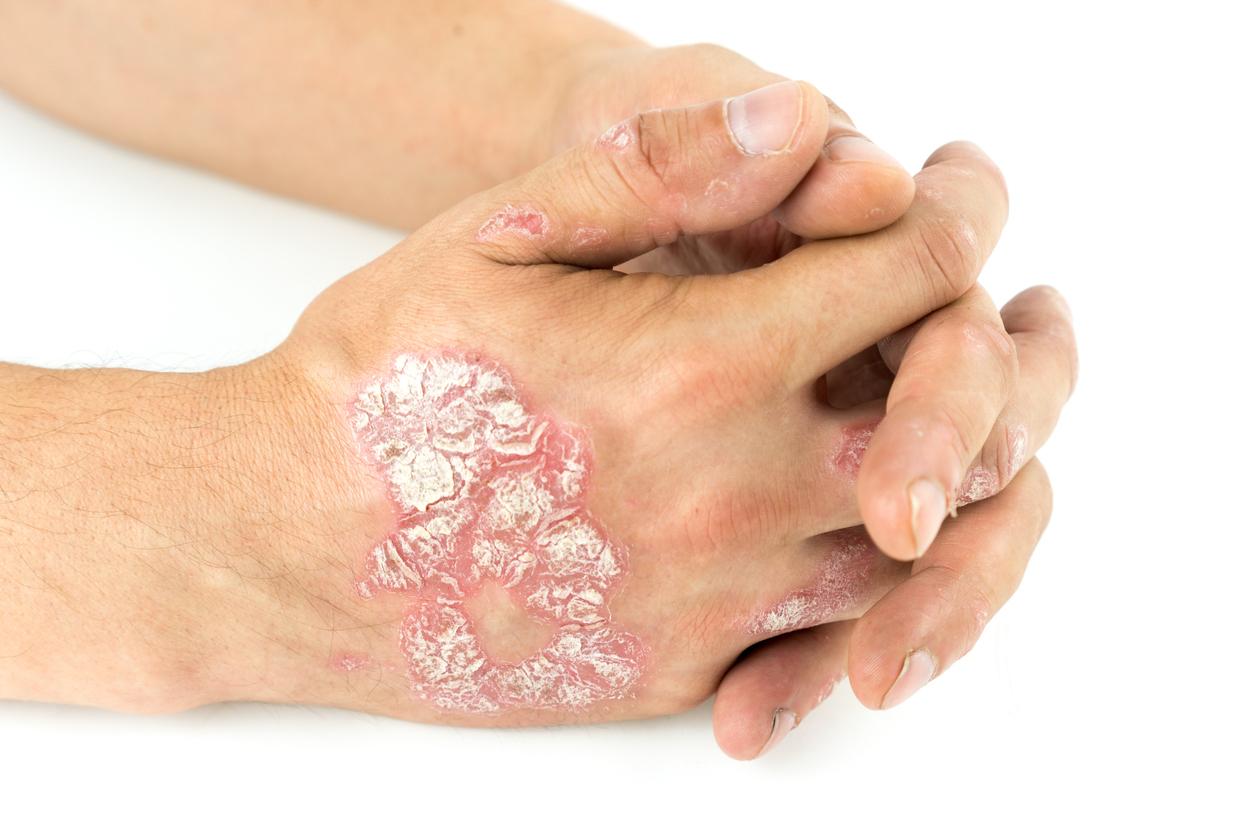
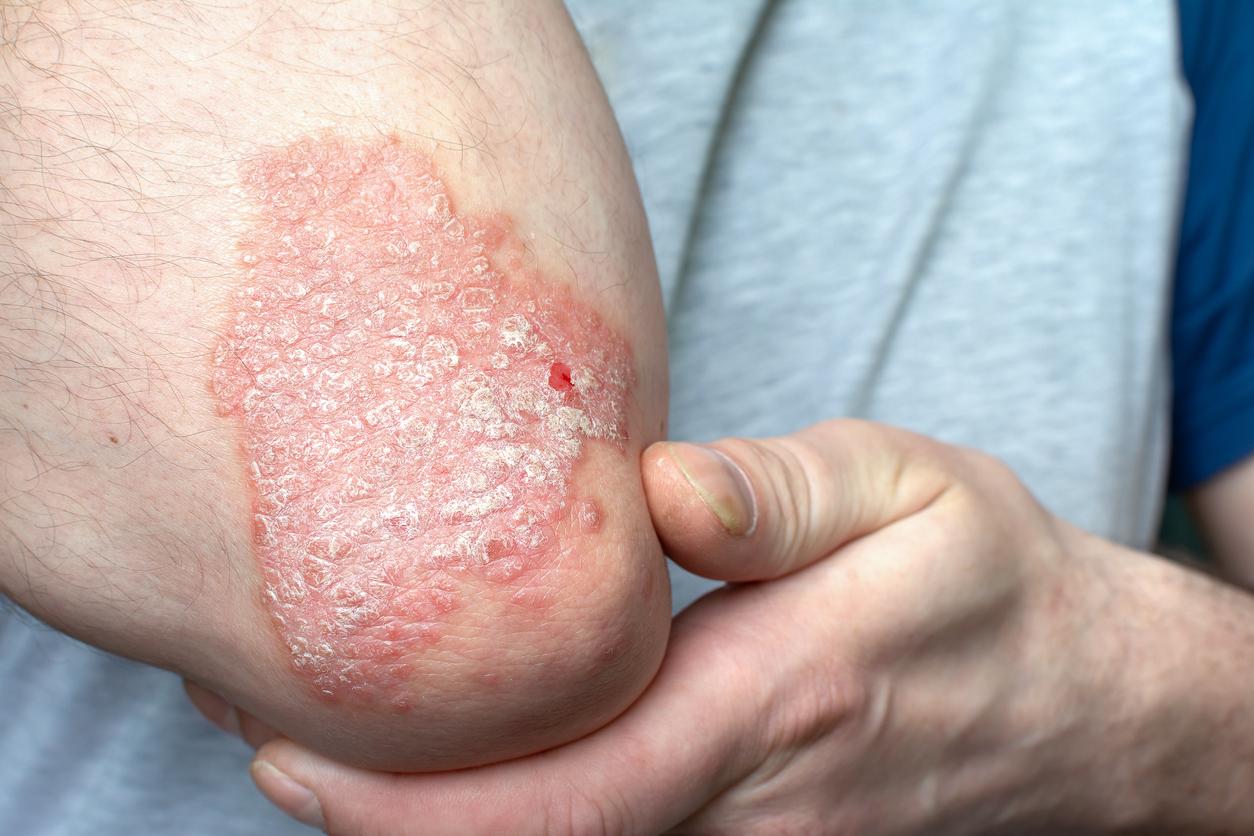
August 29, 2001 – Psoriasis is estimated to affect 2% of the Western population. This incurable disease is not dangerous, but very unpleasant for those who suffer from it. However, two pharmaceutical companies have developed drugs that could cause prolonged remissions after stopping treatment. The news is worth its weight in gold, when we know that the cessation of currently used treatments sometimes causes a rebound effect: psoriasis then resurfaces and worsens.
Amevive® and Remicade®, respectively manufactured by Biogen and Centocor, could be marketed in about a year. At least that’s what Dr. Alice B. Gottlieb said.1 at the world psoriasis conference2 held last June in San Francisco. In fact, the two products have reached phase III of clinical trials, that is to say that which precedes a request for approval from the Food and Drug Administration.
What is unique about these two experimental drugs is that they are expressly designed to target and neutralize part of the immune response associated with the onset of psoriasis. In the case of Amevive (alefacept), some patients have experienced remission lasting up to 18 months, with an average of 10 months. In the case of Remicade (infliximab), half of the subjects who received the drug saw significant improvement in their symptoms for 6 months without having to receive another dose. Remicade is already approved for the treatment of Crohn’s disease and rheumatoid arthritis. Both products were administered by injection to subjects with moderate to severe psoriasis.
The side effects of these drugs are expected to be less severe than those of current products which can cause high blood pressure and even irreversible kidney failure. However, since they suppress part of the immune response, they may, in some patients, increase the risk of serious infections such as tuberculosis as is the case with Remicade used to treat Crohn’s disease and arthritis.3. In clinical trials with Amevive, subjects reported headaches, rash, infections (such as colds), sore throats, and rhinitis, but other side effects could occur over time. term. A major concern for many of those present at the San Francisco conference.
Françoise Ruby – PasseportSanté.net
According to Intelihealth – March 9, 2001
1. Professor of Medicine and Director of the Clinical Research Center at Robert Wood Johnson Medical School, University of Medicine and Dentistry of New Jersey.
2. International Psoriasis Symposium and the European Congress of Psoriasis, 2001 World Conference
3. Centocor Updates REMICADE® (infliximab) Prescribing Information, 08/15/2001