Prime Minister Jean Castex believes that France has entered “a form of third wave”. He is betting on vaccination to get out of it, while the use of the AstraZeneca vaccine is suspended in France and 11 other European countries.

- For Jean Castex, France is currently experiencing a “third wave” of the epidemic
- Vaccination remains the priority in this health crisis, despite the temporary suspension of the AstraZeneca product
- The European Medicines Agency believes about this vaccine that “the benefits far outweigh the risks”
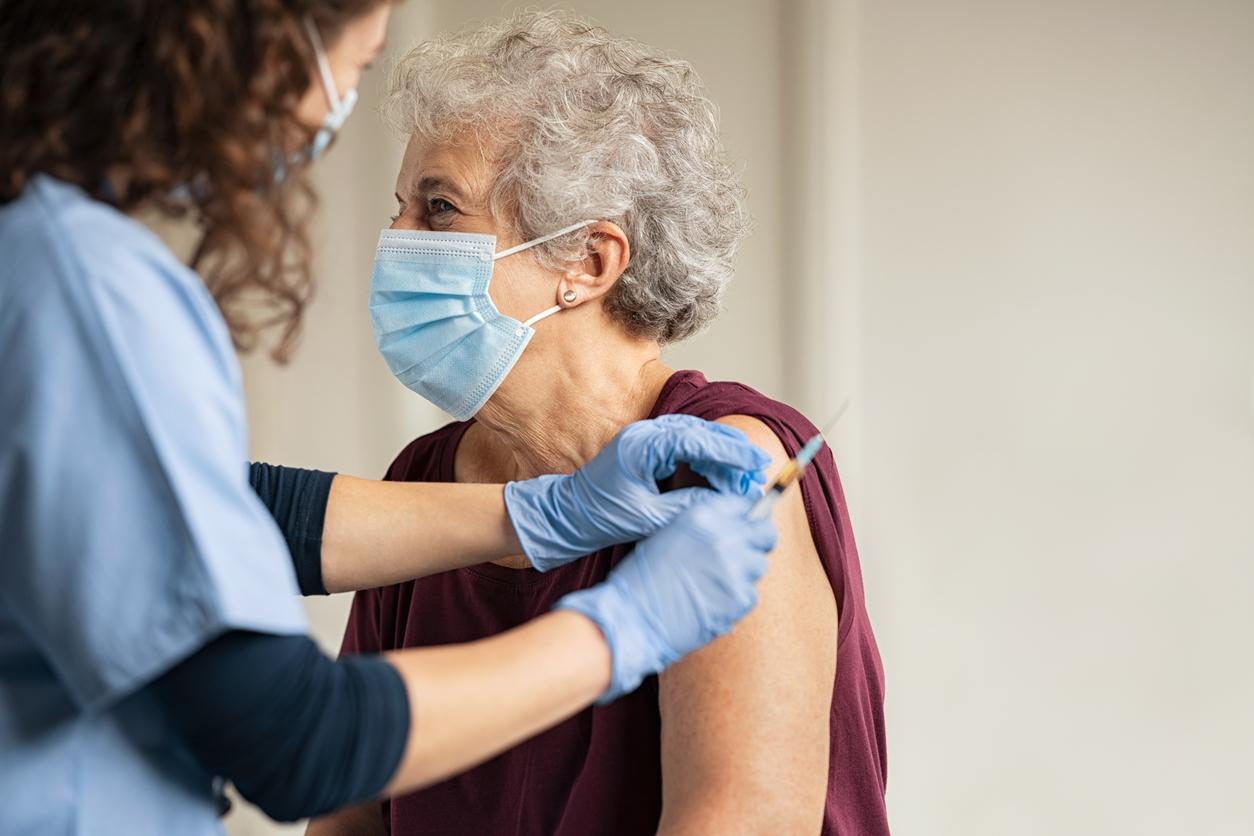
France has entered “a form of third wave of Covid-19 characterized by numerous variants”. This is the formula that many feared, the third wave. These words spoken by Jean Castex on Tuesday afternoon in the hemicycle of the National Assembly could frighten. So, immediately, the Prime Minister tried to reassure. As from the beginning, he repeats, he is betting on vaccination “at the forefront of strategy” to stem the Covid-19 epidemic. According to Public Health France, 5,135,616 people had received at least one dose of the vaccine in France as of March 14, 2021. Nearly 89% of residents in accommodation establishments for dependent elderly people (EHPAD) or in long-term care units (USLD) received, at least, the first injection.
Twelve European countries have suspended AstraZeneca’s vaccine
Problem: if vaccination is the only way out, what will be the effects of the suspension of vaccinations with the AstraZeneca product decided by twelve European governments, including France, in less than a week? After various side effects observed, such as blood clotting disorders, and deaths occurring after the injection of the first dose, the government authorities acted as a precautionary principle. Indeed, for the moment, there is no certainty to establish a link between the vaccine and these side effects. An investigation is underway at European level to determine whether these medical problems are due to AstraZeneca’s product.
“The benefits of AstraZeneca’s vaccine far outweigh the risks”
For its part, AstraZeneca defends its vaccine. “A careful review of all available safety data on over 17 million people vaccinated in the European Union and the UK with (AstraZeneca’s vaccine) showed no evidence of an increased risk of pulmonary embolism, deep vein thrombosis or thrombocytopenia, in any particular age group, sex, lot or country.”, explains the laboratory in a press release published on March 14.
All the European countries which have suspended this vaccine, even if the precautionary principle is justified, are depriving themselves of several thousand doses. This is why some countries like England have chosen not to ban it. Pending future decisions, the European authorities, for their part, seem in favor of continuing vaccination with the AstraZeneca product. According to Ermer Cooke, the director of the European Medicines Agency (EMA), “the benefits of AstraZeneca’s vaccine far outweigh the risks.”
.