He was the fifth to have received a bioprosthetic heart. The transplant patient last August died, confirmed the Carmat company. According to the latter, the prosthesis would not be involved in this death.
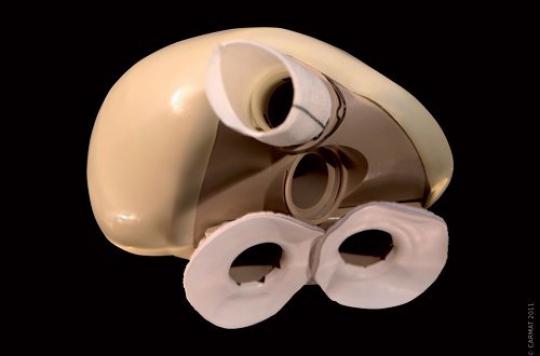
It is not the Carmat company, but the National Medicines Safety Agency (ANSM) which confirmed, this Wednesday evening to several media, the death of the fifth patient to have received an artificial heart. The information was disclosed by Europe 1.
The Carmat company announced on August 29 that it had implanted a new patient. This was the first of the PIVOT trial, the second clinical phase of the project. The man had been operated on at Laënnec hospital in Nantes but no other details had been revealed by Carmat, always very discreet in his communication, since the death of the first implanted patient. The company confirmed, in a press release on the evening of November 30, the death of this patient.
The ANSM would have been informed last October of the death of this fifth patient, according to theExpress. The Agency said it was not aware of the precise date of this death. Following this announcement, the ANSM requested the suspension of the PIVOT trial, in order to determine the causes of this death. Carmat believes for his part that “The analyzes carried out did not show any implication of the prosthesis in the death of the patient”.
During the feasibility study, carried out between late 2013 and early 2016, four patients had been implanted; all were deceased. The patient transplanted in August 2014 survived the longest, 9 months, 4 of which spent at home.
20 patients were to be implanted
Carmat announced on July 13 that it had obtained the green light from the French authorities to start the second phase of its clinical trial, during which 20 patients were to be included in 10 centers in Europe. The main novelty, compared to the feasibility study, is the broadening of the inclusion criteria for patients. Volunteers can now be people eligible for a heart transplant, but having little chance of receiving a heart because at the end of the list. Until now, implantations have only been carried out on patients with end-stage heart failure, who, due to their condition or age, were not eligible for a transplant. Another change in the PIVOT trial: implanted patients must be followed for 180 days, compared to only 30 days in the feasibility study.
“We would like to express our gratitude to this patient and his family. The key contribution made to the CARMAT project by the patients who participated in the clinical trials of the prosthesis is essential in order to offer a lasting solution to patients suffering from advanced heart failure (…) ”, commented Stéphane Piat, Managing Director of CARMAT.
.